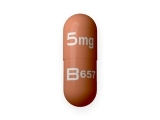
Metformin 1000 mg tablet recall
Important Safety Recall Alert: Do Not Use
Attention all individuals taking Metformin 1000 mg tablets. Our company has issued a voluntary recall of this product due to potential contamination concerns.
This recall is being undertaken as a precautionary measure to ensure your safety and wellbeing.
If you or someone you know has been prescribed Metformin 1000 mg tablets, please discontinue use immediately and consult with your healthcare provider.
Reason for Recall:
Based on our internal quality control processes, we have identified the presence of elevated levels of a potentially harmful substance called N-Nitrosodimethylamine (NDMA).
NDMA is classified as a probable human carcinogen, which means it could increase the risk of cancer for individuals who have been exposed to high levels over long periods of time.
Your safety is our utmost priority, and we apologize for any inconvenience caused by this recall.
If you have any questions or concerns regarding this recall, please contact our customer service hotline at xxx-xxx-xxxx. Our team of trained professionals will assist you in resolving any issues and providing further guidance.
Once again, please discontinue use of Metformin 1000 mg tablets immediately and consult with your healthcare provider.
Thank you for your understanding and cooperation.
Overview of the Recall
The Recall of Metformin 1000 mg Tablets is an important announcement for individuals who have been prescribed this medication for the management of type 2 diabetes. Metformin is commonly prescribed to help control blood sugar levels and is widely used worldwide. However, recent testing has indicated the presence of a potential carcinogen, N-nitrosodimethylamine (NDMA), in certain batches of Metformin 1000 mg Tablets. As a result, the drug manufacturer has initiated a voluntary recall of these specific batches to ensure the safety and well-being of patients.
Understanding the Concern:
The presence of NDMA in medications has raised concerns due to its potential link to an increased risk of cancer. NDMA is a known environmental contaminant found in various sources, including food and water. However, its presence in drugs is not permitted due to the potential health risks it poses. While the levels of NDMA detected in the recalled Metformin 1000 mg Tablets are currently considered to be within acceptable limits, the recall has been initiated as a precautionary measure to prevent any potential harm to patients.
Impacted Batches:
The recall specifically applies to certain batches of Metformin 1000 mg Tablets. These batches can be identified by their specific batch numbers, which can be found on the product packaging. Patients are advised to carefully check the batch number of their medication and compare it to the list provided by the drug manufacturer. If the batch number matches any of those listed, individuals are advised to stop using the medication and contact their healthcare provider for further guidance.
Next Steps for Patients:
If you have been prescribed Metformin 1000 mg Tablets, it is essential to check the batch number of your medication to determine if it is impacted by the recall. If your medication is affected, do not panic. Contact your healthcare provider for guidance on how to proceed. They will be able to provide you with alternative treatment options and ensure that your diabetes management plan is not interrupted. It is important to prioritize your health and work closely with your healthcare team to ensure the best course of action moving forward.
Reasons for the Recall
1. Presence of N-Nitrosodimethylamine (NDMA)
One of the key reasons for the recall of Metformin 1000 mg tablets is the presence of N-Nitrosodimethylamine (NDMA). NDMA is classified as a potential human carcinogen by the International Agency for Research on Cancer (IARC). It is a substance that can be found in certain foods, drinking water, air pollution, and industrial processes. The presence of NDMA above the acceptable daily intake level in Metformin products raises concerns about the potential long-term health risks for patients.
2. Health Risks Associated with NDMA
Exposure to high levels of NDMA over an extended period of time has been linked to an increased risk of cancer in various organs, including the liver, bladder, and stomach. It is important to note that the levels of NDMA found in recalled batches of Metformin 1000 mg tablets exceed the acceptable daily intake level established by regulatory authorities. Therefore, the recall is necessary to protect the health and safety of patients who have been prescribed this medication.
3. Supplier Contamination Issues
The recall of Metformin 1000 mg tablets also stems from supplier contamination issues. The pharmaceutical companies responsible for manufacturing and distributing these tablets have identified potential contamination in certain batches of the active pharmaceutical ingredient (API) used to produce Metformin. This contamination raises concerns about the quality and reliability of the medication, necessitating the recall to prevent any potential harm to patients.
4. Regulatory Compliance and Patient Safety
The recall of Metformin 1000 mg tablets is a proactive measure taken by the pharmaceutical companies to ensure regulatory compliance and prioritize patient safety. By recalling the affected batches, the companies are demonstrating their commitment to adhering to stringent quality control standards and ensuring that patients receive safe and effective medications. The recall also allows for further investigation into the root causes of the contamination and the implementation of appropriate corrective actions to prevent a recurrence in the future.
5. Communication and Public Awareness
An important aspect of the recall is the communication and public awareness surrounding the issue. Pharmaceutical companies, healthcare professionals, and regulatory authorities are working together to inform patients and healthcare providers about the recall, the reasons behind it, and the appropriate steps to take. This ensures that patients are well-informed about the potential risks and can make informed decisions regarding their healthcare. The transparency and open communication surrounding the recall help to maintain trust and confidence in the healthcare system.
Contamination Concerns
Unsafe Ingredients
In recent years, there have been growing concerns about the safety of medications due to the presence of unsafe ingredients. One such medication that has come under scrutiny is Metformin 1000 mg Tablets. It has been found that some batches of this medication may contain unsafe levels of a chemical called N-nitrosodimethylamine (NDMA).
Potential Health Risks
The presence of NDMA in Metformin 1000 mg Tablets raises serious health concerns. NDMA is classified as a probable human carcinogen, meaning that it has the potential to cause cancer. Exposure to high levels of NDMA over a long period of time may increase the risk of developing certain types of cancer, including liver, kidney, and lung cancer.
In addition to the potential cancer risk, exposure to NDMA can also lead to other adverse health effects. Short-term exposure to high levels of NDMA can cause liver damage, while long-term exposure may result in liver fibrosis, scarring, and even liver failure. Other health effects may include respiratory problems and damage to the kidneys and digestive system.
Ensuring Safety and Quality
Ensuring the safety and quality of medications is of utmost importance. The contamination concerns surrounding Metformin 1000 mg Tablets highlight the need for strict adherence to quality control measures throughout the manufacturing process. It is essential that pharmaceutical companies implement thorough testing protocols to detect and prevent the presence of unsafe ingredients in their products.
Consumers also play a crucial role in ensuring their own safety. It is important to stay informed about recalls and product safety alerts, and to follow the guidance of healthcare professionals. If you are currently taking Metformin 1000 mg Tablets or any other medication, consult with your healthcare provider to discuss any concerns you may have and explore alternative treatment options if necessary.
Potential Health Risks
1. Increased Blood Sugar Levels
Consuming Metformin 1000 mg tablets that have been recalled may lead to increased blood sugar levels in patients with diabetes. This can result in poor glycemic control and can have serious consequences for individuals who rely on medication to manage their blood sugar levels.
2. Adverse Effects on Cardiovascular Health
Using the affected Metformin tablets may pose risks to cardiovascular health. Patients with pre-existing heart conditions may experience exacerbation of their symptoms or increased risk of cardiovascular events, such as heart attack or stroke. It is important to discontinue use of the recalled tablets and consult a healthcare professional immediately.
3. Potential Contamination
The recall of Metformin 1000 mg tablets is due to potential contamination with a potential carcinogen called N-Nitrosodimethylamine (NDMA). This compound has been classified as a probable human carcinogen and may increase the risk of cancer over time. It is crucial to stop using the recalled tablets and seek alternative treatment options.
4. Impaired Liver Function
Continued use of the affected Metformin tablets may lead to impaired liver function. The liver plays a crucial role in metabolizing drugs, and any disruption can lead to adverse health effects. Patients who have been using the recalled tablets should consult a healthcare professional for further guidance and monitoring of their liver function.
5. Potential Digestive Upset
Some patients may experience digestive upset, such as nausea, vomiting, or diarrhea, after consuming the recalled Metformin tablets. It is important to discontinue use of the affected tablets and report any adverse effects to a healthcare professional for proper evaluation and management.
6. Possible Allergic Reactions
Individuals who are allergic to the active ingredient in Metformin or any other components of the tablets may experience allergic reactions. These can range from mild symptoms, such as itching and hives, to severe reactions, including difficulty breathing and swelling of the face, lips, or tongue. If any allergic reactions occur, immediate medical attention should be sought.
It is crucial to prioritize your health and well-being. If you have been using Metformin 1000 mg tablets that have been recalled, it is important to discontinue use and consult with a healthcare professional for guidance on alternative treatments.
Impact on Consumers
1. Possible Adverse Effects
Consumers who have been taking Metformin 1000 mg tablets may be at risk of experiencing adverse effects due to the recall. These adverse effects can include gastrointestinal problems such as nausea, vomiting, and diarrhea. It is important for consumers to be aware of these potential issues and to consult with their healthcare provider if they experience any of these symptoms.
2. Disruption of Treatment
The recall of Metformin 1000 mg tablets can also have a significant impact on consumers' treatment plans for managing their diabetes. If consumers are unable to access their medication due to the recall, it may lead to a disruption in their treatment and potentially affect their blood sugar control. It is crucial for consumers to stay updated on the recall and to work with their healthcare provider to find alternative medications or treatment options.
3. Financial Costs
Another factor that can affect consumers is the financial cost associated with the recall. If consumers need to switch to a different medication or treatment option, it may result in additional expenses. Furthermore, if consumers have already purchased Metformin 1000 mg tablets that are part of the recall, they may have to bear the cost of returning or disposing of the medication. This can have a financial burden on individuals who rely on the medication to manage their diabetes.
4. Anxiety and Concern
The recall of a medication can also cause anxiety and concern among consumers. They may worry about the safety and efficacy of the medication they have been taking, especially if they have been relying on it for a long time. Consumers may also feel uncertain about the availability of alternative medications or treatment options. It is important for healthcare providers to address these concerns and provide reassurance to patients affected by the recall.
5. Need for Information and Support
Consumers impacted by the recall will likely have a need for accurate and up-to-date information regarding the recall, as well as guidance on how to manage their diabetes during this period. They may also require emotional support to deal with the potential disruption and uncertainty. It is crucial for healthcare providers and support organizations to provide accessible information and resources to assist consumers during this time of recall.
Availability of Alternative Medications
1. Metformin Extended-Release Tablets
If you are currently taking Metformin 1000 mg Tablets and are looking for an alternative medication, Metformin Extended-Release Tablets may be an option for you. These tablets provide the same active ingredient, metformin hydrochloride, but in an extended-release formula. This means that the medication is released slowly over time, providing a steady level of the drug in your body. Metformin Extended-Release Tablets are available in various strengths, including 500 mg, 750 mg, and 1000 mg, so you can find the dosage that works best for you.
2. Metformin Combination Products
Another alternative medication option is Metformin Combination Products. These medications combine metformin with other drugs that help to control blood sugar levels. For example, some combination products may include metformin and a dipeptidyl peptidase-4 (DPP-4) inhibitor, or metformin and a sodium-glucose cotransporter-2 (SGLT2) inhibitor. These combination products provide the benefits of metformin along with additional medications that can further help to regulate blood sugar levels.
3. Insulin Therapy
In cases where oral medications are not enough to control blood sugar levels, insulin therapy may be necessary. Insulin is a hormone that helps to regulate blood sugar levels in the body. It can be administered through injections or an insulin pump. If you are unable to take metformin or other oral medications, or if they are not effectively managing your blood sugar, your healthcare provider may recommend insulin therapy as an alternative treatment option.
It is important to discuss your options with your healthcare provider to determine the best alternative medication for your specific needs. They can evaluate your medical history, current medications, and individual health goals to make an informed recommendation. Remember, every individual is different, and what works for one person may not be the best option for another.
Lack of Medical Treatment
Are you tired of the lack of medical treatment options for your condition? Look no further! Our innovative solution is here to provide you with the personalized care you deserve.
Customized Treatment Plans
At Lack of Medical Treatment, we understand that every individual is unique, and so are their healthcare needs. That's why we offer customized treatment plans tailored to your specific condition. Our team of experienced doctors will work closely with you to develop a plan that addresses your concerns and improves your overall well-being.
Advanced Medical Technologies
With our state-of-the-art medical technologies, we can accurately diagnose and treat a wide range of conditions. Whether you're dealing with chronic pain, a complex illness, or a rare disease, our advanced equipment and techniques ensure that you receive the highest level of care. You can trust us to stay at the forefront of medical advancements in order to provide you with the most effective treatments available.
Comprehensive Care
We believe that comprehensive care is essential for optimal health outcomes. That's why our team of specialists, including doctors, surgeons, and therapists, work together to provide you with a holistic approach to treatment. From diagnosis to recovery, we will be by your side, ensuring that your medical needs are met every step of the way.
Don't settle for subpar medical treatment. Contact Lack of Medical Treatment today and experience the difference that personalized care can make in your life.
Steps Taken by the Manufacturer
The manufacturer of the metformin 1000 mg tablets has taken several steps to address the recall and ensure the safety of the consumers. Firstly, they have notified healthcare professionals and wholesalers about the recall, providing them with detailed information about the affected lots and potential risks.
Additionally, the manufacturer has conducted a thorough investigation to determine the root cause of the issue and has implemented corrective actions to prevent similar incidents in the future. They have reviewed their manufacturing processes and quality control procedures to identify any potential weaknesses and make necessary improvements.
To further ensure the safety of the consumers, the manufacturer is working closely with regulatory authorities to address the recall and comply with all necessary requirements. They are collaborating with the FDA and other agencies to ensure that the affected products are removed from the market and replaced with safe alternatives.
The manufacturer has also established a dedicated customer support line to assist consumers and healthcare professionals with any questions or concerns regarding the recall. They are committed to providing prompt and accurate information to help address any potential issues and ensure the well-being of the patients.
In conclusion, the manufacturer of the metformin 1000 mg tablets has taken swift and decisive steps to address the recall and safeguard the health of the consumers. They have notified relevant parties, conducted investigations, implemented corrective actions, and collaborated with regulatory authorities. Additionally, they have provided dedicated customer support to address any concerns. Their commitment to ensuring product safety is paramount, and they will continue to take necessary measures to uphold this commitment.
Follow us on Twitter @Pharmaceuticals #Pharmacy
Subscribe on YouTube @PharmaceuticalsYouTube




Be the first to comment on "Metformin 1000 mg tablet recall"